Communication between cancer cells and surrounding stromal cells is essential for tumor progression. Our study revealed the existence of a “vicious circle” between the tumor and the stroma favoring the progression of acute myeloid leukemia (AML).
In our previous work, we identified a syntenin-dependent signaling pathway that regulates cell-cell communication. Syntenin controls the recycling and presence at the plasma membrane of numerous receptors, as well as the production and uptake of small extracellular vesicles. Numerous studies to date suggest that overexpression of syntenin in tumor cells promotes tumor aggressiveness, making it a promising target for cancer therapy. Several syntenin inhibitors, including those we have developed at CRCM, have shown promising results in preclinical cancer models. However, the specific role of syntenin in the tumor microenvironment remains poorly understood.
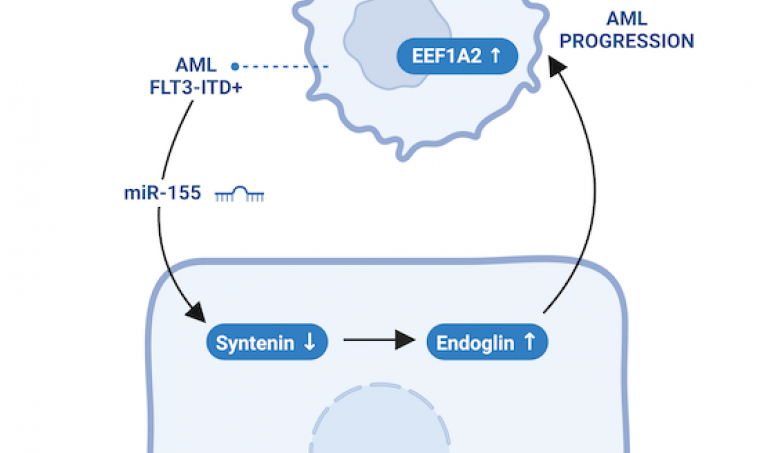
By consulting a public database, we discovered that, surprisingly, syntenin expression is reduced in bone marrow stromal cells (BMSCs) isolated from leukemia (AML) mice. Consistent with this observation, we demonstrated that leukemia cells are indeed able to decrease syntenin expression in BMSCs by up to threefold, by transferring a microRNA (miR-155), which is overexpressed in 30% of human AML cases.
We have also demonstrated that a significant decrease in syntenin expression in BMSCs has pro-tumor effects, suggesting that there is a “feedback loop” between leukemia cells and stromal cells. Using serial transplantation experiments in mice expressing or not syntenin, and in vitro co-culture experiments, we were able to demonstrate that the absence of syntenin in BMSCs stimulates the aggressiveness of leukemia cells, promoting their survival, and that this effect is due to an accumulation of a glycoprotein, endoglin, on the surface of BMSCs. There is ample evidence to suggest that high levels of endoglin on stromal cells promote tumor progression. However, the way in which endoglin on stromal cells contributes to this progression has until now been poorly characterized.
In summary, our data reveal an unsuspected “tumor suppressor” effect of syntenin, calling into question for the first time the relevance and safety of using syntenin as a target in cancer therapy. This is true in the context of AML, but also potentially in tumours arising in bone.
We are particularly proud to point out that this study was carried out entirely at the CRCM, thanks to a fruitful collaboration involving the TrGET platform, the proteomics platform, Michel Aurrand-Lions’ team, and clinicians at the Institut Paoli-Calmettes.
